|
For the rest of this discussion though, I’ll be focusing solely on the Brønsted-Lowry theory of acids and bases. According to the Lewis theory, an acid is an electron pair acceptor, while a base is an electron pair donor.įor the purposes of this lesson, I am not going to go any further into the details of the Lewis theory and leave it for another lesson. His acid-base theory is sometimes called the electronic theory of acids and bases. There’s one last even more general acid base theory proposed by the American physical chemist Gilbert Lewis. 
But in this particular case, the HSO 4 – ion is a conjugate base. The conjugate base that we made in the reaction above (HSO 4 –) can dissociate further and be an acid in a different reaction. 
Remember, that when we are talking about the conjugates we are always talking about the products of a specific acid-base reaction. 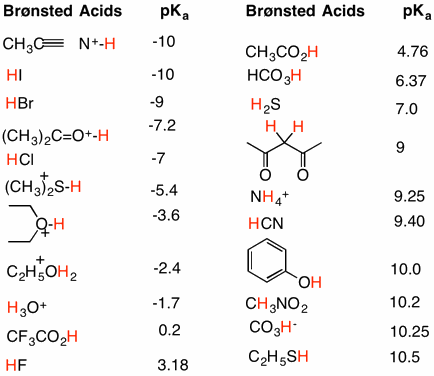
And once a base accepts a proton, it becomes a conjugate acid. Thus, once an acid loses the proton, it becomes a conjugate base.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
